Reference: Ferrea, E., Negahbani, F., Cebi, I., Weiss, D., & Gharabaghi, A. (2024). Machine learning explains response variability of deep brain stimulation on Parkinson’s disease quality of life. npj Digital Medicine, 7, Article 269.
Imagine living with Parkinson’s disease and coping with symptoms ranging from tremors and muscle stiffness to problems with balance and thinking. Your doctor offers a brain surgery that could substantially improve your motor symptoms, but warns that it may not relieve other challenges, such as difficulty with daily life activities, depression, or concentrating on tasks. Deciding whether to proceed would not be easy. You might weigh which symptoms impact your quality of life the most, knowing that the surgery likely can’t fix everything.
One treatment option for Parkinson’s disease is Deep Brain Stimulation (DBS), a procedure in which surgeons implant an electrode in the brain to stimulate neural activity. Although DBS is highly invasive, it can reduce motor symptoms by 50% or more in many patients (2). Improvements in quality-of-life outcomes, however, are less predictable. Outcomes can vary depending on factors such as a patient’s age, how long they have had the disease, and their individual brain anatomy. In the study discussed here, researchers found that the strongest predictor of improved quality of life after DBS was a specific pattern of brain activity near the implanted electrode. The best predictor of worsening quality of life, somewhat paradoxically, was having a relatively higher quality-of-life before surgery.
To investigate these outcomes, researcher Ferrea and colleagues recruited 63 patients undergoing DBS surgery and measured their quality of life both before and after the surgery. Among the participants, 34 (54%) reported an improvement in quality of life, while 28 (46%) of patients experienced worsening outcomes quality of life or no change. The researchers aimed to determine which of more than a dozen patient characteristics best predicted an improvement or decline in quality-of-life score. To address the uncertainty surrounding DBS outcomes, they applied machine learning (a form of artificial intelligence), which is particularly useful for identifying patterns across large, complex datasets.
Quality of Life in Parkinson’s Disease
Parkinson’s disease (PD) is a progressive neurodegenerative disorder characterized by motor symptoms such as tremor, slowed movements (bradykinesia), and muscle rigidity. These symptoms arise when key communication pathways in the brain begin to deteriorate. Although motor changes are the most visible features of PD, many people with the disease also experience a wide range of non-motor symptoms. These can include sleep difficulties, depression and anxiety, and problems with impulse control. Together, the combination of motor and non-motor challenges can substantially reduce quality of life for people living with PD, especially as the disease progresses.
Many strategies can help manage PD symptoms and slow their progression without surgery, such as lifestyle adjustments, exercise, and medication. DBS is a more invasive treatment option, but it can provide long-lasting improvements in motor symptoms and may reduce the need for medication. On the other hand, effects on non-motor symptoms and on overall quality of life as the disease progresses are less clear. This uncertainty likely contributes to the variability researchers observe in patients’ quality-of-life outcomes after DBS surgery.
To evaluate diverse outcomes more systematically, researchers often use the Parkinson’s Disease Questionnaire (PDQ-39). This assessment has 39 questions covering eight different domains of daily life, such as mobility, activities of daily living, emotional well-being, and cognition. Scores from these domains are combined to create an overall measure of quality of life, with lower scores indicating better perceived quality of life.
Machine Learning Can Predict Outcomes Based on Individual Characteristics
Why might DBS lead to improvements in quality-of-life outcomes for some people but worse outcomes for others? To investigate this question, the researchers collected a wide range of information about each participant. These inputs included clinical measures such as the patient’s pre-surgery PDQ-39 score, as well as basic information like age, sex, and disease duration. The dataset also included neural data collected during surgery, including the precise three-dimensional location of the implanted electrode and the strength of brain activity recorded around it.
Using these inputs, the researchers trained a machine learning model to predict the patient’s PDQ-39 scores after surgery. The model used a decision tree-based strategy, a method commonly applied across fields such as healthcare and finance (see Figure 1 below). In simple terms, the decision tree models repeatedly split data based on the variables that best separate outcomes. For this study, the model also improved over time by learning from errors in previous prediction attempts, iteratively refining its ability to forecast post-surgery quality-of-life outcomes.
After training the model, the researchers examined which variables contributed most strongly to the predictions. Two factors emerged as the most important: patients’ pre-surgery PDQ-39 scores and the strength of a specific type of brain activity measured during surgery. Surprisingly, lower pre-surgery PDQ-39 scores, which reflect better quality of life, were associated with worse quality-of-life scores after surgery. This pattern may reflect the progressive nature of PD: even if surgery improves motor symptoms, quality of life may still decline over time as other symptoms worsen. It may also suggest that DBS is less effective for certain symptoms that strongly influence quality of life.
The best predictor of improved quality-of-life was the power of high beta oscillations recorded near the electrode. Neural oscillations are patterns of rhythmic activity within the brain that are thought to reflect communication across neural networks. In Parkinson’s disease, beta oscillations (often divided into “low” and “high” based on the frequency of their rhythm) have been studied extensively. Low beta activity in particular has been linked to motor symptoms (3), and both low and high beta oscillations can decrease in strength following DBS treatment (see Figure 2).
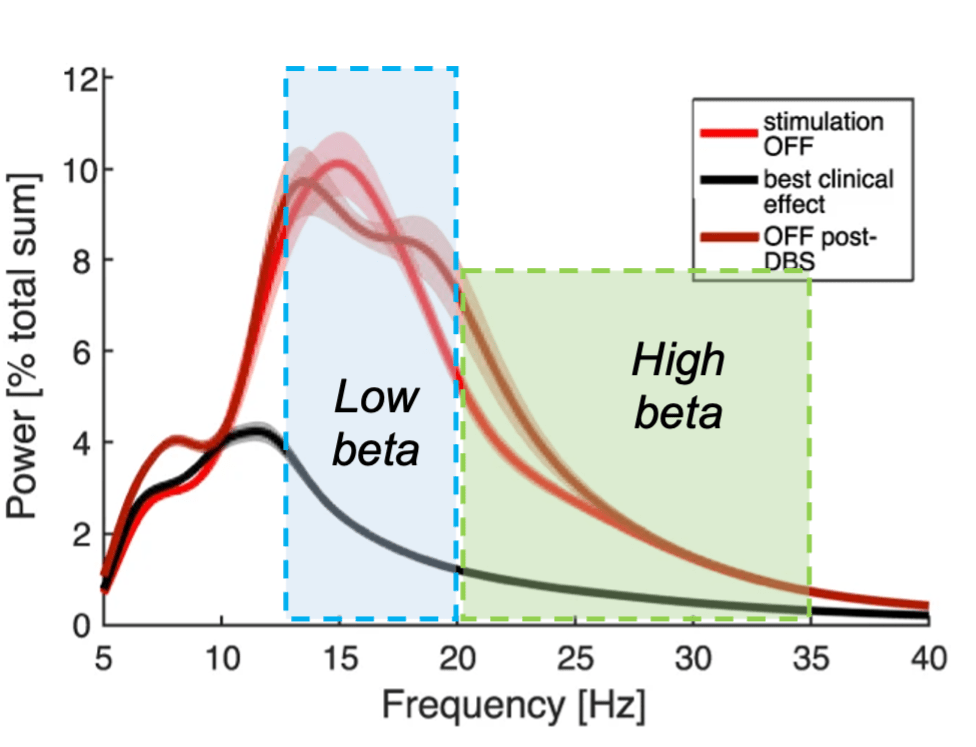
Far less is known about the behavioral or clinical symptoms that may be associated with high beta activity. However, because high beta power was the strongest predictor of quality-of-life improvements, the researchers speculate that Deep Brain Stimulation may be influencing neural circuits beyond those involved in motor control. In other words, DBS might have broader therapeutic potential for networks contributing to emotional, cognitive, or daily functioning outcomes.
Other factors (such as patients’ age and the final location of the implanted electrode) were considerably less important for the model’s predictions. However, they still provided useful information when combined with other variables, helping the machine learning model generate more accurate predictions overall.
How Can this Study Inform More Effective DBS Treatment?
It is important to consider this study only looks at a single (albeit wholistic) measure of quality of life and examines neural activity within one specific brain target. Even with these limitations, the findings highlight two major factors that could help improve DBS treatment outcomes in people with Parkinson’s disease: careful patient selection and the growing promise of intraoperative biomarkers (neural signals measured during surgery).
One particularly unexpected result was that patients with lower pre-surgery quality-of-life burden were more likely to experience worse outcomes after Deep Brain Stimulation. This result suggests that clinicians may need to more carefully consider which patients are the best candidates for surgery. For example, individuals with milder quality-of-life impairment might benefit more from alternative therapies, depending on the specific symptoms and progression pattern of their disease.
At the same time, the mixed quality-of-life outcomes observed in this study and previous research highlight an ongoing challenge: current treatments for Parkinson’s disease are still far better at addressing motor symptoms than non-motor symptoms. The importance of high beta brain activity as a predictor of positive outcomes suggests that DBS has promise to improve more than just motor symptoms, even if the mechanisms are still unclear. Understanding these neural signals could eventually help clinicians tailor stimulation settings or surgical targeting to improve patient outcomes.
Additionally, the machine learning framework used for this study can be applied to other common DBS targets, not only in Parkinson’s disease but also in other disorders that benefit from DBS. By identifying the factors that predict successful outcomes, researcher and clinicians can move closer to understanding why DBS works and how we can make it work better for even more people.
Additional References:
- Feldmann, L. K., Lofredi, R., Neumann, W. J., Al-Fatly, B., Roediger, J., Bahners, B. H., Nikolov, P., Denison, T., Saryyeva, A., Krauss, J. K., Faust, K., Florin, E., Schnitzler, A., Schneider, G. H., & Kühn, A. A. (2022). Toward therapeutic electrophysiology: beta-band suppression as a biomarker in chronic local field potential recordings. NPJ Parkinson’s disease, 8(1), 44. https://doi.org/10.1038/s41531-022-00301-2
- Lachenmayer, M. L., Mürset, M., Antih, N., Debove, I., Muellner, J., Bompart, M., Schlaeppi, J. A., Nowacki, A., You, H., Michelis, J. P., Dransart, A., Pollo, C., Deuschl, G., & Krack, P. (2021). Subthalamic and pallidal deep brain stimulation for Parkinson’s disease-meta-analysis of outcomes. NPJ Parkinson’s disease, 7(1), 77. https://doi.org/10.1038/s41531-021-00223-5
- Radcliffe, E. M., Baumgartner, A. J., Kern, D. S., Al Borno, M., Ojemann, S., Kramer, D. R., & Thompson, J. A. (2023). Oscillatory beta dynamics inform biomarker-driven treatment optimization for Parkinson’s disease. Journal of neurophysiology, 129(6), 1492–1504. https://doi.org/10.1152/jn.00055.2023
Images:
- Featured photo by Andreas Horn from Wikimedia Commons (CC-BY 4.0)
- Image from Abhishek Sharma on Geeks for Geeks. Accessed Feb 10 2026. Link.
